
Site saturation mutagenesis (SSM), or simply site saturation, is a random mutagenesis technique used in protein engineering, in which a single codon or set of codons is substituted with all possible amino acids at the position.[1] There are many variants of the site saturation technique, from paired site saturation (saturating two positions in every mutant in the library) to scanning site saturation (performing a site saturation at every site in the protein, resulting in a library of size [20 x (number of residues in the protein)] that contains every possible point mutant of the protein).
Method
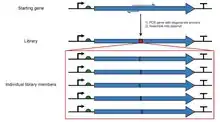
Saturation mutagenesis is commonly achieved by site-directed mutagenesis PCR with a randomised codon in the primers (e.g. SeSaM)[2] or by artificial gene synthesis, with a mixture of synthesis nucleotides used at the codons to be randomised.[3]
Different degenerate codons can be used to encode sets of amino acids.[1] Because some amino acids are encoded by more codons than others, the exact ratio of amino acids cannot be equal. Additionally, it is usual to use degenerate codons that minimise stop codons (which are generally not desired). Consequently, the fully randomised 'NNN' is not ideal, and alternative, more restricted degenerate codons are used. 'NNK' and 'NNS' have the benefit of encoding all 20 amino acids, but still encode a stop codon 3% of the time. Alternative codons such as 'NDT', 'DBK' avoid stop codons entirely, and encode a minimal set of amino acids that still encompass all the main biophysical types (anionic, cationic, aliphatic hydrophobic, aromatic hydrophobic, hydrophilic, small).[1] In the case there is no restriction to use a single degenerate codon only, it is possible to reduce the bias considerably.[4][5] Several computational tools were developed to allow high level of control over the degenerate codons and their corresponding amino acids.[6][7][8]
| Degenerate codon | No. of codons | No. of amino acids | No. of stops | Amino acids encoded |
|---|---|---|---|---|
| NNN | 64 | 20 | 3 | All 20 |
| NNK / NNS | 32 | 20 | 1 | All 20 |
| NDT | 12 | 12 | 0 | RNDCGHILFSYV |
| DBK | 18 | 12 | 0 | ARCGILMFSTWV |
| NRT | 8 | 8 | 0 | RNDCGHSY |
Applications
Saturation mutagenesis is commonly used to generate variants for directed evolution.[9][10]
See also
References
- 1 2 3 Reetz, M. T.; Carballeira J. D. (2007). "Iterative saturation mutagenesis (ISM) for rapid directed evolution of functional enzymes". Nature Protocols. 2 (4): 891–903. doi:10.1038/nprot.2007.72. PMID 17446890. S2CID 37361631.
- ↑ Zheng, Lei; Baumann, Ulrich; Reymond, Jean-Louis (2004-07-15). "An efficient one-step site-directed and site-saturation mutagenesis protocol". Nucleic Acids Research. 32 (14): e115. doi:10.1093/nar/gnh110. ISSN 0305-1048. PMC 514394. PMID 15304544.
- ↑ Reetz, Manfred T.; Prasad, Shreenath; Carballeira, José D.; Gumulya, Yosephine; Bocola, Marco (2010-07-07). "Iterative Saturation Mutagenesis Accelerates Laboratory Evolution of Enzyme Stereoselectivity: Rigorous Comparison with Traditional Methods". Journal of the American Chemical Society. 132 (26): 9144–9152. doi:10.1021/ja1030479. ISSN 0002-7863. PMID 20536132.
- ↑ Kille, Sabrina; Acevedo-Rocha, Carlos G.; Parra, Loreto P.; Zhang, Zhi-Gang; Opperman, Diederik J.; Reetz, Manfred T.; Acevedo, Juan Pablo (2013-02-15). "Reducing Codon Redundancy and Screening Effort of Combinatorial Protein Libraries Created by Saturation Mutagenesis". ACS Synthetic Biology. 2 (2): 83–92. doi:10.1021/sb300037w. PMID 23656371.
- ↑ Tang, Lixia; Wang, Xiong; Ru, Beibei; Sun, Hengfei; Huang, Jian; Gao, Hui (June 2014). "MDC-Analyzer: a novel degenerate primer design tool for the construction of intelligent mutagenesis libraries with contiguous sites". BioTechniques. 56 (6): 301–302, 304, 306–308, passim. doi:10.2144/000114177. ISSN 1940-9818. PMID 24924390.
- ↑ Halweg-Edwards, Andrea L.; Pines, Gur; Winkler, James D.; Pines, Assaf; Gill, Ryan T. (September 16, 2016). "A Web Interface for Codon Compression". ACS Synthetic Biology. 5 (9): 1021–1023. doi:10.1021/acssynbio.6b00026. ISSN 2161-5063. PMID 27169595.
- ↑ Engqvist, Martin K. M.; Nielsen, Jens (2015-04-30). "ANT: Software for Generating and Evaluating Degenerate Codons for Natural and Expanded Genetic Codes". ACS Synthetic Biology. 4 (8): 935–938. doi:10.1021/acssynbio.5b00018. PMID 25901796.
- ↑ Kell, Douglas B.; Day, Philip J.; Breitling, Rainer; Green, Lucy; Currin, Andrew; Swainston, Neil (2017-07-10). "CodonGenie: optimised ambiguous codon design tools". PeerJ Computer Science. 3: e120. doi:10.7717/peerj-cs.120. ISSN 2376-5992.
- ↑ Chica, Robert A.; et al. (2005). "Semi-rational approaches to engineering enzyme activity: combining the benefits of directed evolution and rational design". Current Opinion in Biotechnology. 16 (4): 378–384. doi:10.1016/j.copbio.2005.06.004. PMID 15994074.
- ↑ Shivange, Amol V; Marienhagen, Jan; Mundhada, Hemanshu; Schenk, Alexander; Schwaneberg, Ulrich (2009-02-01). "Advances in generating functional diversity for directed protein evolution". Current Opinion in Chemical Biology. Biocatalysis and Biotransformation/Bioinorganic Chemistry. 13 (1): 19–25. doi:10.1016/j.cbpa.2009.01.019. PMID 19261539.