 | |
 | |
| Names | |
|---|---|
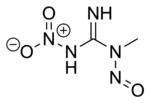
| Preferred IUPAC name
N-Methyl-N′-nitro-N-nitrosoguanidine | |
| Other names
1-Methyl-3-nitro-1-nitrosoguanidine N-Methyl-N-nitroso-N′-nitroguanidine | |
| Identifiers | |
3D model (JSmol) |
|
| Abbreviations | MNNG |
| ChemSpider | |
| ECHA InfoCard | 100.000.664 |
| KEGG | |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H5N5O3 | |
| Molar mass | 147.09 g/mol |
| Appearance | Yellow crystals |
| Melting point | 118 °C (244 °F; 391 K) (decomposes) |
| reacts violently, slowly hydrolysed | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Methylnitronitrosoguanidine (MNNG[2] or MNG, NTG when referred to colloquially as nitrosoguanidine[3]) is a biochemical tool used experimentally as a carcinogen and mutagen.[1] It acts by adding alkyl groups to the O6 of guanine and O4 of thymine, which can lead to transition mutations between GC and AT. These changes do not cause a heavy distortion in the double helix of DNA and thus are hard to detect by the DNA mismatch repair system.
One of the earliest uses of methylnitronitrosoguanidine was in 1985. A group of scientists tested whether or not the chemical composition of methylnitronitrosoguanidine would directly affect the growth of tumors and cancer cells in rats.
In the experiment, the cancer cells from a Japanese cancer patient was injected into 8 rats. The biochemical tool and showed a decline of cancer cells in a few of the rats' bodies.
In organic chemistry, MNNG is used as a source of diazomethane when reacted with aqueous potassium hydroxide.[4]
MNNG is a probable human carcinogen listed as an IARC Group 2A carcinogen.[5]
Stability
MNNG produces diazomethane (known DNA methylating agent) in basic aqueous solutions, and nitrous acid (also mutagenic) in acidic solutions.[6]
References
- 1 2 Merck Index, 11th Edition, 6017.
- ↑ Lawley, P. D.; Thatcher, Carolyn J. (1970-02-01). "Methylation of deoxyribonucleic acid in cultured mammalian cells by N-methyl-N′-nitro-N-nitrosoguanidine. The influence of cellular thiol concentrations on the extent of methylation and the 6-oxygen atom of guanine as a site of methylation". Biochemical Journal. Portland Press Ltd. 116 (4): 693–707. doi:10.1042/bj1160693. ISSN 0306-3283. PMC 1185415. PMID 5435496.
- ↑ "Forward and reverse mutagenesis in C. elegans". WormBook. Retrieved 2021-12-01.
- ↑ T. Howard Black (1983). "The Preparation and Reactions of Diazomethane" (PDF). Aldrichimica Acta. 16 (1). Archived from the original (PDF) on 2019-02-03. Retrieved 2019-02-02.
- ↑ N-METHYL-N'-NITRO-N-NITROSOGUANIDINE (MNNG), International Agency for Research on Cancer
- ↑ Hollaender, Alexander (1971). Chemical Mutagens : Principles and Methods for Their Detection Volume 1. Boston, MA: Springer US. p. 188. ISBN 978-1-4615-8966-2. OCLC 851813793.