 | |
| Clinical data | |
|---|---|
| Trade names | Flucort |
| Other names | Flumethasone (USAN) |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Liver, CYP3A4-mediated |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.016.701 |
| Chemical and physical data | |
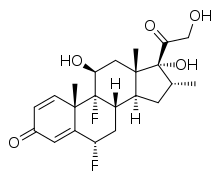
| Formula | C22H28F2O5 |
| Molar mass | 410.458 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Flumetasone, also known as flumethasone,[1] is a corticosteroid for topical use.
It was patented in 1951 and approved for medical use in 1964.[2]
Chemistry
Flumethasone is 420 times as potent as cortisone in an animal model for anti-inflammatory activity.
Society and culture
Names
Trade names include Locacorten, Locorten, and Orsalin. It is available in combination with clioquinol, under the brand name Locacorten-Vioform (in some countries Locorten-Vioform), for the treatment of otitis externa and otomycosis. It is usually formulated as the pivalic acid ester prodrug called flumetasone pivalate.
Veterinary uses
In April 2023, the US Food and Drug Administration approved the first generic version of flumethasone (Bimasone), for certain diseases that cause inflammation in horses, dogs, and cats.[3] Flumetasone is recommended for the various rheumatic, allergic, dermatologic and other disease states that are known to be responsive to anti-inflammatory drugs (i.e., corticoids) in these species.[3]
In horses, flumetasone is approved for use for musculoskeletal conditions due to inflammation, such as bursitis (a painful condition that affects the fluid-filled sacs that cushion the bones, tendons and muscles near a horse’s joints, often in the hip or shoulder), carpitis (inflammation of the connective tissues near the horse’s carpus, which is comparable to the human wrist), osselets (arthritis in the fetlock joint of a horse, often in the front legs), and myositis (rapid muscle wasting, often in the horse’s hindquarters).[3]
In dogs, Bimasone is approved for use for musculoskeletal conditions due to inflammation of muscles or joints, such as arthritis, osteoarthritis, intervertebral disc disease, and myositis.[3] The drug has proven useful in treating canine ear infections (otitis externa) when used with topical medication for similar reasons.[3] It is also approved for certain acute and chronic skin conditions (dermatoses) from various causes to help control the itchy skin (pruritus), irritation, and inflammation associated with these conditions, and for use in treating allergic reactions, such as hives, urticaria (raised itchy rash on skin), and insect bites.[3] Flumetasone is also approved for treating shock in dogs, when administered intravenously.[3]
In cats, flumetasone is approved for certain acute and chronic skin conditions (dermatoses) from various causes to help control the itchy skin (pruritus), irritation, and inflammation associated with these conditions.[3]
References
- ↑ Morton IK, Hall JM (1999). Concise Dictionary of Pharmacological Agents Properties and Synonyms. Dordrecht: Springer Netherlands. p. 122. ISBN 9789401144391.
- ↑ Fischer J, Ganellin CR (2006). Analogue-based Drug Discovery. John Wiley & Sons. p. 484. ISBN 9783527607495. Archived from the original on 10 January 2023. Retrieved 7 April 2023.
- 1 2 3 4 5 6 7 8 "FDA Approves Generic Flumethasone for Diseases in Cats, Dogs and Horse". U.S. Food and Drug Administration (FDA). 5 April 2023. Archived from the original on 7 April 2023. Retrieved 7 April 2023.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.