 | |
| Clinical data | |
|---|---|
| Trade names | Fluonex, Lidex, others[1] |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a601054 |
| Routes of administration | Topical |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | hepatic |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.998 |
| Chemical and physical data | |
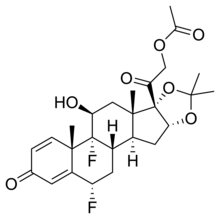
| Formula | C26H32F2O7 |
| Molar mass | 494.532 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Fluocinonide (sold under various brand names) is a potent glucocorticoid used topically as an anti-inflammatory agent for the treatment of skin disorders such as eczema and seborrhoeic dermatitis.[3] It relieves itching, redness, dryness, crusting, scaling, inflammation, and discomfort.[3]
A common potential adverse effect is skin atrophy (thinning of the skin).[4] Systemic absorption of topical corticosteroids can produce reversible hypothalamic-pituitary-adrenal axis (HPA) suppression, manifestations of Cushing's syndrome, hyperglycemia, and glucosuria.
In 2020, it was the 280th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[5][6]
Veterinary uses
Fluocinonide is used in veterinary medicine. It is a treatment for allergies in dogs.[7] Natural systemic cortisol concentrations can be suppressed for weeks after one week of topical exposure.[8]
References
- ↑ "Fluocinonide Topical". MedlinePlus Drug Information. U.S. National Library of Medicine.
- ↑ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- 1 2 "Fluocinonide Topical: MedlinePlus Drug Information". medlineplus.gov. Retrieved 2022-11-16.
- ↑ Jablonska S, Groniowska M, Dabroswki J (February 1979). "Comparative evaluation of skin atrophy in man induced by topical corticoids". The British Journal of Dermatology. 100 (2): 193–206. doi:10.1111/j.1365-2133.1979.tb05561.x. PMID 154921. S2CID 30656827.
- ↑ "The Top 300 of 2020". ClinCalc. Retrieved 7 October 2022.
- ↑ "Fluocinonide - Drug Usage Statistics". ClinCalc. Retrieved 7 October 2022.
- ↑ Dog Allergies. Squidoo (Report).
- ↑ Zenoble RD, Kemppainen RJ (September 1987). "Adrenocortical suppression by topically applied corticosteroids in healthy dogs". Journal of the American Veterinary Medical Association. 191 (6): 685–688. PMID 2824410.
External links
- "Fluocinonide". Drug Information Portal. U.S. National Library of Medicine.