| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1,1,1-Trichloro-2-methylpropan-2-ol | |||
| Other names
1,1,1-Trichloro-2-methyl-2-propanol; Chlorbutol; Chloreton; Chloretone; Chlortran; Trichloro-tert-butyl alcohol; 1,1,1-Trichloro-tert-butyl alcohol; 2-(Trichloromethyl)propan-2-ol; tert-Trichlorobutyl alcohol; Trichloro-tert-butanol; Trichlorisobutylalcohol; 2,2,2-Trichloro-1,1-dimethylethanol | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.000.288 | ||
| EC Number |
| ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C4H7Cl3O | |||
| Molar mass | 177.45 g·mol−1 | ||
| Appearance | White solid | ||
| Odor | Camphor | ||
| Melting point | 95–99 °C (203–210 °F; 368–372 K) | ||
| Boiling point | 167 °C (333 °F; 440 K) | ||
| Slightly soluble | |||
| Solubility in acetone | Soluble | ||
| Pharmacology | |||
| A04AD04 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Xn | ||
| NFPA 704 (fire diamond) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
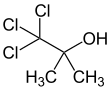
Chlorobutanol (trichloro-2-methyl-2-propanol) is an organic compound with the formula CCl3C(OH)(CH3)2. The compound is an example of a chlorohydrin. The compound is a preservative, sedative, hypnotic and weak local anesthetic similar in nature to chloral hydrate. It has antibacterial and antifungal properties.[1] Chlorobutanol is typically used at a concentration of 0.5% where it lends long term stability to multi-ingredient formulations. However, it retains antimicrobial activity at 0.05% in water. Chlorobutanol has been used in anesthesia and euthanasia of invertebrates and fishes.[2][3] It is a white, volatile solid with a camphor-like odor.
Synthesis

Chlorobutanol was first synthesized in 1881 by the German chemist Conrad Willgerodt (1841–1930).[4]
Chlorobutanol is formed by the reaction of chloroform and acetone in the presence of potassium or sodium hydroxide. It may be purified by sublimation or recrystallisation.[5]
Parthenogenesis
Chlorobutanol has proven effective at stimulating parthenogenesis in sea urchin eggs up to the pluteus stage, possibly by increasing irritability to cause stimulation. For the eggs of the fish Oryzias latipes, however, chlorobutanol only acted as an anesthetic.[6]
Pharmacology and toxicity
It is an anesthetic with effects related to isoflurane and halothane.[7]
Chlorobutanol is toxic to the liver, a skin irritant and a severe eye irritant.[8]
References
- ↑ Noecker, Robert (2001). "Effects of common ophthalmic preservatives on ocular health". Advances in Therapy. 18 (5): 205–215. doi:10.1007/bf02853166. PMID 11783457. S2CID 1455107.
- ↑ W. N. McFarland & G. W. Klontz (1969). "Anesthesia in fishes". Federation Proceedings. 28 (4): 1535–1540. PMID 4894939.
- ↑ John E. Cooper (2011). "Anesthesia, analgesia, and euthanasia of invertebrates". ILAR Journal. 52 (2): 196–204. doi:10.1093/ilar.52.2.196. PMID 21709312.
- ↑ See:
- Willgerodt, C. (1881). "Ueber die Einwirkung ätzender Alkalien auf halogenirte Verbindungen in Acetonlösungen" [On the action of caustic alkalies on halogenated compounds in acetone solutions]. Berichte der Deutschen Chemischen Gesellschaft (in German). 14 (2): 2451–2460. doi:10.1002/cber.188101402191.
- Willgerodt, C. (1882). "Ueberführung des Acetonchloroforms in die Oxyisobuttersäure" [Conversion of chlorobutanol into 2-hydroxyisobutyric acid]. Berichte der Deutschen Chemischen Gesellschaft (in German). 15 (2): 2305–2308. doi:10.1002/cber.188201502176.
- Willgerodt, C. (1883). "Zur Kenntniss des Acetonchloroforms" [[Contribution] to [our] knowledge of chlorobutanol]. Berichte der Deutschen Chemischen Gesellschaft (in German). 16 (2): 1585. doi:10.1002/cber.18830160218.
- Willgerodt, C.; Genieser, Ad. (1888). "Beiträge zur Kenntniss des Acetonchloroforms" [Contributions to [our] knowledge of chlorobutanol]. Journal für praktische Chemie (in German). 37 (8): 361–374. doi:10.1002/prac.18880370131.
- ↑ "Chlorobutanol". Sciencemadness Wiki. Retrieved 14 August 2020.
- ↑ Embryologia 1956
- ↑ Nicholas P. Franks (2006). "Molecular targets underlying general anaesthesia". British Journal of Pharmacology. 147 (Suppl 1): S72–S81. doi:10.1038/sj.bjp.0706441. PMC 1760740. PMID 16402123.
- ↑ MSDS